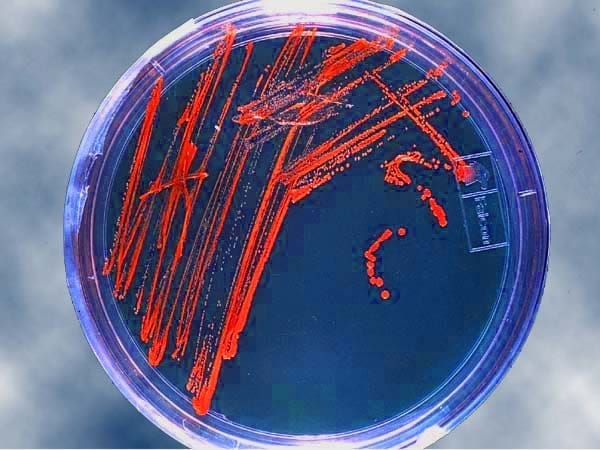
WEST LAFAYETTE, Ind. – Royer Corp. and Poly Group LLC finalized an exclusive partnership June 22 that results in the manufacture of antimicrobial custom plastic injection molded parts in Madison, Indiana.
Nouvex antimicrobial technology was discovered at Purdue University by a group led by Jeffrey Youngblood, a professor in Purdue’s School of Materials Engineering. Poly Group LLC holds an exclusive, worldwide license to the IP covering Nouvex, which includes a granted U.S. patent.
“It is rewarding to see our technology go to the market and help make a difference, particularly during this worldwide pandemic,” Youngblood said.
Royer Corp., a Madison-based manufacturer, serves a variety of industries that will benefit from an antimicrobial option – especially given the new safety challenges businesses are facing because of the COVID-19 pandemic.
Royer will infuse Nouvex material in a variety of custom injection molded applications such as airline and mass transit interior parts, food service and hospitality components/disposables, cosmetic applicators, housewares and commercial product pieces.
“Given our company’s unique injection molding capabilities, virtually any custom plastic product can be made in antimicrobial material,” said Roger Williams, CEO of Royer.
The antimicrobial technology marketed as Nouvex has been licensed exclusively and globally to Poly Group by the Purdue Research Foundation Office of Technology Commercialization.
The office is now housed in the Convergence Center for Innovation and Collaboration in Discovery Park District, adjacent to the Purdue campus.
About Poly Group LLC
Poly Group LLC is located in the Purdue Research Park in New Albany, Indiana. The antimicrobial technology marketed as Nouvex has been licensed exclusively and globally to Poly Group by the Purdue Research Foundation Office of Technology Commercialization. Poly Group has successfully registered Nouvex with the U.S. Environmental Protection Agency (EPA), as a material preservative; EPA Reg. No. 91413-2.
About Purdue Research Foundation Office of Technology Commercialization
The Purdue Research Foundation Office of Technology Commercialization operates one of the most comprehensive technology transfer programs among leading research universities in the U.S. Services provided by this office support the economic development initiatives of Purdue University and benefit the university’s academic activities through commercializing, licensing and protecting Purdue intellectual property. The office recently moved into the Convergence Center for Innovation and Collaboration in Discovery Park District, adjacent to the Purdue campus. In fiscal year 2019, the office reported 136 deals finalized with 231 technologies signed, 380 disclosures received and 141 issued U.S. patents. The office is managed by the Purdue Research Foundation, which received the 2019 Innovation and Economic Prosperity Universities Award for Place from the Association of Public and Land-grant Universities. In 2020, IPWatchdog Institute ranked Purdue third nationally in startup creation and in the top 20 for patents. The Purdue Research Foundation is a private, nonprofit foundation created to advance the mission of Purdue University.
Contact Poly Group LLC
For more information on the science behind Nouvex and ways you can help bring this solution to market faster, contact Craig Kalmer, Chief Operating Officer, Poly Group LLC at (812) 590-4750.
Image Credit
Cover image courtesy of Royer Corporation.